Research
LIVER
Nonalcoholic fatty liver disease (NAFLD) is a major health care challenge with a global prevalence of approximately 25%. Numbers up to 90% are reported in high-risk groups suffering from severe obesity and type 2 diabetes mellitus. Patients with NAFLD have an increased overall mortality, for which the most common cause is [cardiovascular disease]. NAFLD is increasingly recognized as the liver disease component of the metabolic syndrome. NAFLD represents a spectrum of disorders and starts with lipid accumulation in the liver (i.e. steatosis), but it may progress to nonalcoholic steatohepatitis (NASH), characterized by inflammation with hepatocyte injury (ballooning) with or without fibrosis, which may eventually result in cirrhosis. A liver biopsy or MRI/MRS still are the gold standards for detection and staging of steatosis, however these techniques are expensive and invasive, and in biopsies complication such as bleeding and infection may occur. Aim: We aim to predict the liver fat content non-invasively using 2D ultrasound images in a generic fashion, such that this technique can used on all ultrasound modalities consisting a digital image output.
Research aim
Concerning the high prevalence of steatosis in combination with the lag of symptons and serious risk on the progression of steatosis into hepatitis, fibrosis and cirrhosis, we aim to develop a low-cost, generic and non-invasive ultrasound technique to identify steatosis, such that it can be used for screening and follow-up purposes.
Methods
Steatosis
We developed and tested the CAlibrated UltraSound (CAUS) protocol for the assessment of liver fat content (i.e. steatosis), using calibrated conventional 2D ultrasound (B-mode) images. CAUS is generic reference phantom method, which determines various quantitative ultrasound parameters semi-automatically like the ”residual attenuation coefficient (RAC, [dB.cm-1.MHz-1])”, “Mean echo level (MU, [dB])”, and parametric “texture features" like the axial and lateral speckle size and amplitudes. CAUS expresses the echo level parameters relatively to a (commercial available) reference phantom, and compensated for the echo level depth dependency (i.e. the beam profile) which results in depth independent measure of the RAC and relative echo levels. This makes CAUS generic applicable to all ultrasound modalities, consisting a digital image interface/output and to various applications.
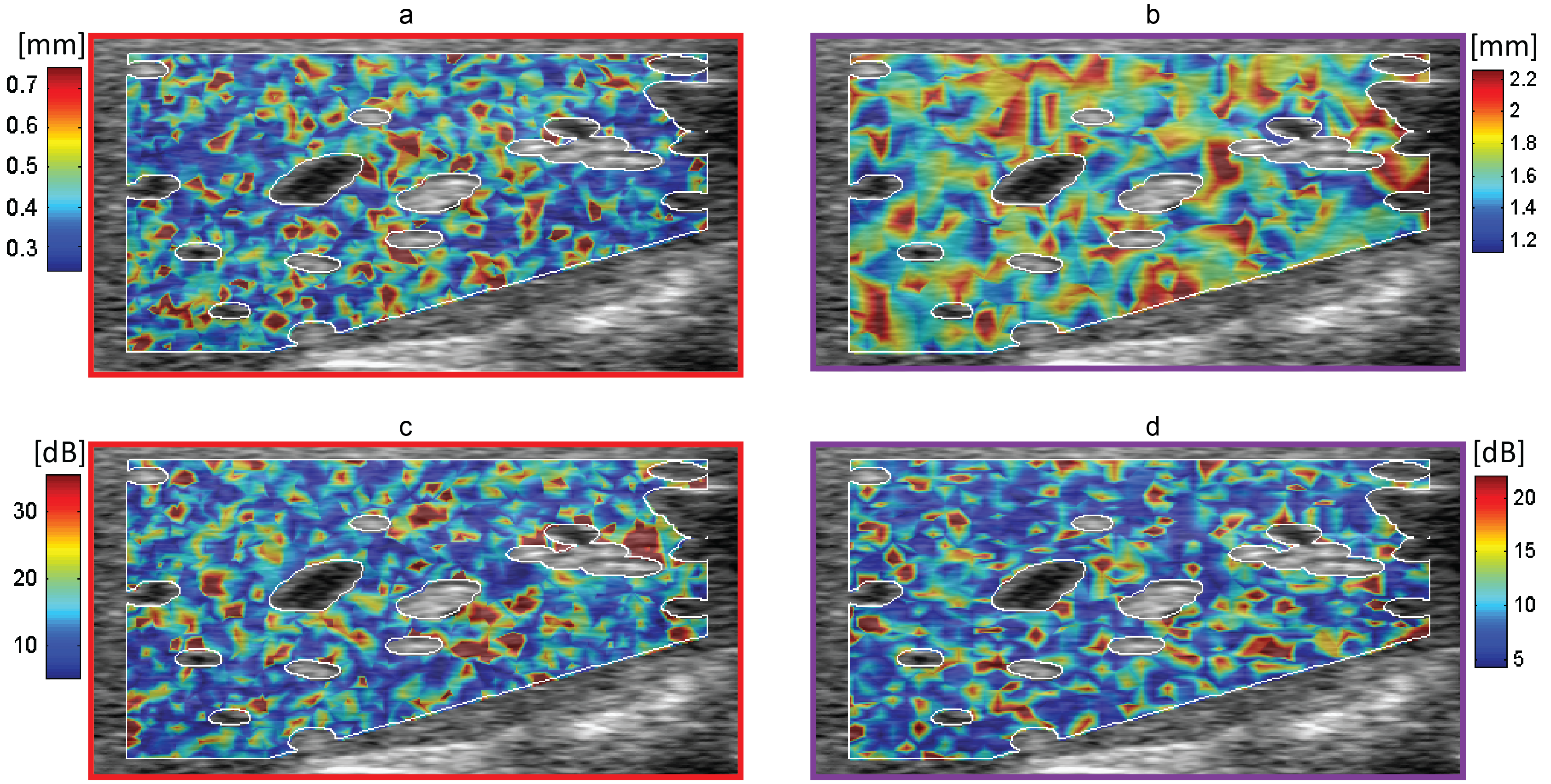
Figure 1. Parametric texture images of (a) Axial speckle size (b) Lateral speckle size (c) Axial speckle amplitude (d) Lateral speckle amplitude
Fibrosis
For detection and staging of fibrosis several commercial ultrasound techniques like: Transient Elestography (TE) and Shear wave elastography (SWE) are available. We currently are comparing the inter-changeability of those techniques, as well as comparing the Residual Attenuation Coefficient (RAC) from CAUS vs the Continues Attenuation Parameter (CAP) of the Fibroscan device in a large cohort of HIV patients (n=2000).
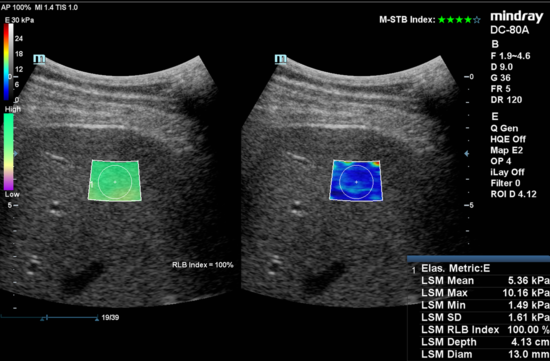
Figure 2. Example image of clinically obtained liver stiffness by using Shear Wave Elastography (Mindray Medical)
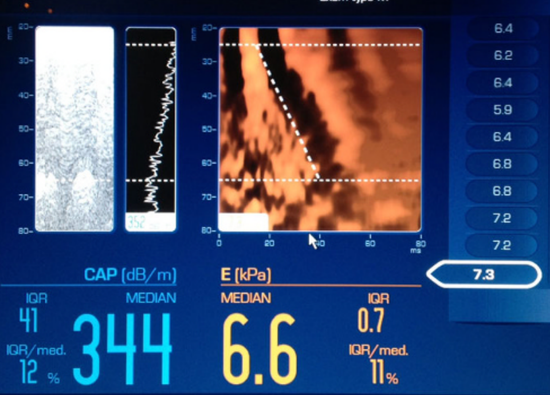
Figure 3. Fibroscan example image with left: echo level curve from which the CAP [dB/m] is estimated, and right: shear wave propagation map from which the stiffness [kPa] is derived.
Results
CAUS showed high predictive values (AUROC up to 0.95) in bovine studies (Weijers et al. 2012, 2010, 2010a, Thijsen et al. 008) and in a human pilot study confirmed similar performance of CAUS for the detection of hepatic steatosis, compared to magnetic resonance spectroscopy (Weijers et al. 2016).
To validate these positive results a large human validation study (n=214) was performed in colaboration with the Gastroenterology department to test the diagnostic performance of CAUS for staging hepatic steatosis using a liver biopsy as gold standard, which recently is published in Radiology (Weijers et al. Radiology 2022). In this study even higher predictive performances compared to the cows studies were found for the detection (AUROC: 0.97) and grading (moderated; severe respectively, AUROC: 0.93; 0.93) of steatosis using the RAC parameter only.
Funding
CAUS development is performed without any form of external funding.
Currently a CAUS validation and comparison study is performed within the 2000HIV study, which is funded by ViiV Healthcare.